The Basics[]
According to Electron-pair repulsion theory charge centres will move to be as far apart as possible.
The furthest apart three charge centres can be is 120o.
If all charge centres are all bonds (or double bonds) the shape will be referred to as Trigonal Planar.
Nearly all examples will be Group III elements (B or Al) bonded to H,F,Cl,or Br. Or possibly Alkenes or SO3.
If one charge centre was a lone pair then the bonds would make a V-shape and the additional repulsion of the lone-pair decreases the bond angle to around 118o - but this is not going to be on the exam unless its sulphur trioxide.

3 charge centres, all bonds= Trigonal planar (120o)

3 charge centres, one is a lone pair = V-shape (118o)
Exam Hint[]
- Standard examples of trigonal planar molecules include BF3 and BCl3 etc, AlF3 and AlCl3 etc.
- However,an examiner may throw in some unfamiliar example such as SO3 or an alkene so it is better to be able to work out the shape.
To work out if your molecule really is trigonal planar follow the steps below:
- Which is the central atom?
- Identify how many valence (outer) electrons it has from its group number. (Generally, if it's trigonal it will be a group 3 element -B or Al- but it may be a Sulphur compound or an alkene)
- How many bonds will the other elements form to the central atom? (Generally, 1 for Halogens and Hydrogen, Oxygen forms 2 but this would have to bond to Sulphur which has six electrons available to form three double bonds).
- Each bond formed uses up one of the valence electrons. Are there any left?
- If there are two bonds (or double bonds) and two spare electrons then there is a lone pair. It won't be trigonal planar, it will be v-shape or bent.
- If there are three bonds (or double bonds) and four spare electrons then there are two lone pairs. It won't be trigonal planar but this will not be on the exam.
- In the case of trigonal planar molecules, there must be three (single or double) bonds and no lone-pairs.

Sulphur has 6 electrons free to bond allowing 6 bonds. But 3 double bonds is still 3 charge centres so still trigonal planar.
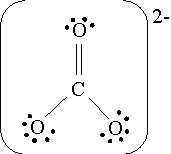
You could be asked about an ion. Extra electrons allow extra bonds. This negative ion is still trigonal planar even though one charge centre is a double bond.

